Vaccines are an Important Component of Dairy Cattle Health Management Programs
Vaccines are an Important Component of Dairy Cattle Health Management Programs
Vaccines stimulate the immune system of dairy cows, calves, and heifers to produce antibodies against a particular disease organism. When the animal is challenged or encounters the cause of the disease at a later time, the animal’s body is able to recognize the disease pathogen and more quickly defend the animal’s body against it. Vaccines help by decreasing the likelihood of dairy cattle getting a disease and/or decrease the severity of the disease. Thus, vaccines are an important and vital part of preventative health programs. They should be viewed as insurance against a potential disease threat. Hopefully, you will never need its defenses, but if needed the defenses are there to help prevent potential health disasters. I still can remember from early in my career, a farm that was not up-to-date on their BVD vaccine, got BVD in the herd, and lost a “major” number of cows and not “just a couple”; all of which could have been prevented with proper vaccination and other health protocols.
In order to get the immune response intended, one needs to not only use the proper vaccines, but they need to be administered properly. Vaccines are prepared from either killed viruses or bacteria (known as killed vaccines) or by modifying a live virus or bacteria so that it no longer causes disease symptoms, but results in an immune response (known as modified live vaccines). Choosing the proper types and form of vaccines for your dairy herd is a conversation you need to have with your local herd veterinarian, since they need to be selected to reflect the diseases found in your herd and local area and the intended animal (calf, heifer or cow). However, using these vaccines properly on your farm is under your, the dairy farm manager’s, control. Missteps can render these vaccines ineffective. Take time to review the proper care and use with those helping administer vaccines on your farm. These areas include the following often forgotten details when administering vaccines.
Prior to Vaccinating Animals
- Read the label insert before administering any vaccines and do this at least each time you purchase a new vial of vaccine. Changes can occur in the dosage of product needed per animal or a different manufacturer of a vaccine may have different directions regarding its proper use.
- Some vaccines, particularly calf vaccines for pneumonia, are administered through the intra-nasal route of delivery.
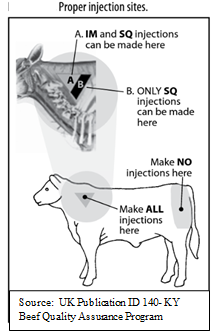
- When the insert gives a choice between an IM or sub-Q injection method, the sub-Q route should be used. All injections, unless specifically noted in the insert directions, should be administered in the neck region. Injections in the flank area result in muscle damage and reductions in carcass quality. Remember that dairy cows have two occupations in their lifetimes, dairy and beef cows.
- Check expiration dates on vaccines before administrating. Expired vaccines may not be effective.
Choose the Correct Needle and Injection Site
- Single use needles are preferred to prevent the spread of blood-borne diseases, such as BLV (Bovine Leukosis Virus), a common disease on dairies.
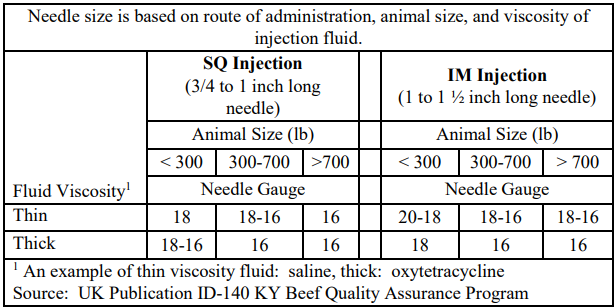
- Use the proper length and gauge of needle dependent on the size of animal, route of administration, and thickness of injected liquid. For sub-Q injections, a ¾ to 1 inch needle should be used versus an 1 to 1 ½ inch long needle for IM injections. When injections are greater than 10 cc, no more than 10 cc should be injected per site with sub-Q injections separated by at least 5 inches.
- After use, needles should be disposed of properly to prevent others from getting accidently stuck by the needle. Sharps containers (or liquid detergent bottles) can be used and disposed as solid waste with the lid attached or cover closed.
Improper Handling Can Make Vaccines Ineffective
- If a vaccine requires boosters or multiple vaccine doses, make sure they are completed on the schedule as described. Failing to do so can decrease the vaccines effectiveness or prevent the immunity response needed. An electronic or wall-hanging calendar can be used to determine when next doses should be given and to what animal(s). The PCDart program from DHI contains a feature that will help remind mangers to complete these tasks.
- Do not store vaccines on the door of the refrigerator. Wide fluctuations in temperature are possible if stored on the door as the refrigerator is opened and closed to retrieve products stored inside. Also, the door is the warmest area of the refrigerator.
- Do not reconstitute too much vaccine at one time. Modified live vaccines begin to degrade after about 1 hour in the heat and sunlight. Reconstituted vaccines (especially modified live vaccines) should be kept in a cooler with an ice pack. Place the ice pack under bubble wrap or a clean towel to prevent freezing of the vaccine.
- When reconstituting vaccines, a new disposable needle and syringe should be used. If a transfer needle is used, it should be cleaned and sterilized by boiling and not by using isopropanol or rubbing alcohol. Using alcohol denatures modified live vaccines, rendering them ineffective.
- To mix the liquid (diluent) with the freeze-dried powder after reconstituting, gently rock the vial back and forth to mix. Do not use a shaking motion like used to mix flavored milk in a plastic jug!
- Vaccines, antibiotics, and other drugs should not be stored with the needle or needle and syringe inserted into the top rubber cap. This practice allows a means for bacteria to enter the vial and cause contamination.
Don’t Forget To Record Vaccinations
- Maintain written or electronic records of the product used, date administered, animals vaccinated/treated, person completing the task, and withdrawal/withholding times for both meat and milk for at least 2 years. Should questions arise regarding the use of antibiotics or vaccines, the FDA may request to see these retrievable records at a later date. Notes made on a dry eraser boards are not available at a later date, if needed.
Authors: Donna M. Amaral-Phillips, Ph.D. and Michelle Arnold, DVM
